Research
- English
- Türkçe
Deregulation in metabolic pathways contribute significantly to cancer progression, not simply by meeting the energy needs of a rapidly growing cell population, but also to provide essential metabolites that can help the tumor cells cope with a rapidly changing microenvironment. Using wet lab and data mining, we aim to better understand signal transduction pathways related to enzymes of lipid and glucose metabolism pathways.
1. Lipid metabolism pathways in colorectal cancer
Eicosanoids are derived from the metabolism of arachidonic acid by enzymes called cyclooxygenases and lipoxygenases. 15-Lipoxygenase-1, a member of this family of enzymes that preferentially metabolize linoleic acid (an essential fatty acid) into 13-hydroperoxy octadecadienoic acid (13-HpODE). The latter can bind to the orphan nuclear receptor PPARγ and induce changes in proliferation, apoptosis, cellular motility, vascularization and inflammation.
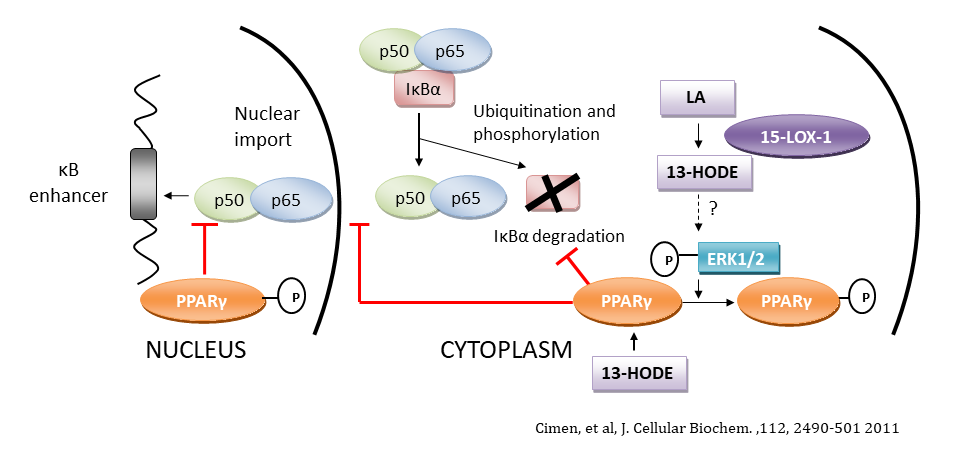
2. Glucose metabolism pathways in colorectal cancer
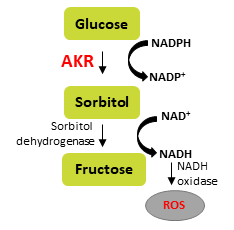 Aldo keto reductases are enzymes of the polyol pathway that are responsible for the conversion of excess glucose into sorbitol with the consumption of NADPH and are therefore strongly implicated in pathologies related to diabetes. We and others have shown that these enzymes are also important in colorectal cancer. AKR1B1 was functional in increasing proliferation, motility, high ROS production and activation of NF-κB, whereas a closely related member, AKR1B10 was more tumor suppressive and was associated with a reduction in inflammatory markers. Interestingly, AKR1B1 high tumors showed poor prognosis whereas AKR1B10 high tumors showed favorable prognosis. A combination of AKR1B10 high and AKR1B1 low tumors show the best prognosis, indicating that these genes could serve as potential biomarkers.
Aldo keto reductases are enzymes of the polyol pathway that are responsible for the conversion of excess glucose into sorbitol with the consumption of NADPH and are therefore strongly implicated in pathologies related to diabetes. We and others have shown that these enzymes are also important in colorectal cancer. AKR1B1 was functional in increasing proliferation, motility, high ROS production and activation of NF-κB, whereas a closely related member, AKR1B10 was more tumor suppressive and was associated with a reduction in inflammatory markers. Interestingly, AKR1B1 high tumors showed poor prognosis whereas AKR1B10 high tumors showed favorable prognosis. A combination of AKR1B10 high and AKR1B1 low tumors show the best prognosis, indicating that these genes could serve as potential biomarkers.
3. Signal transduction during cellular differentiation
The process of cellular differentiation in the gut is highly complex and provides clues not only on the proper functioning of the gut and its barrier function, but also potential signaling mechanisms that contribute towards tumorigenesis of these rapidly proliferating cells. We have shown that intestinal epithelial cells undergo autophagy in the course of differentiation, which may contribute to a reduction in inflammation.
